Antoine-Laurent Lavoisier
Chemist and public servant, b. 26 August 1743 (Paris, France), d. 8 May 1794 (Paris).
 Lavoisier was the son of a parliamentary counsel, who could afford to give his son an excellent classical education in language, literature, and philosophy combined with best available training in the sciences, including mathematics, astronomy, chemistry, and botany. Lavoisier entered university to study law and received his licence to practice in 1764. His main interest, however, was science. In 1766 he submitted an entry to the competition organized by the Academy of Sciences on the best ways to illuminate a large town and received a gold medal.
Lavoisier was the son of a parliamentary counsel, who could afford to give his son an excellent classical education in language, literature, and philosophy combined with best available training in the sciences, including mathematics, astronomy, chemistry, and botany. Lavoisier entered university to study law and received his licence to practice in 1764. His main interest, however, was science. In 1766 he submitted an entry to the competition organized by the Academy of Sciences on the best ways to illuminate a large town and received a gold medal.
Lavoisier became wealthy through his family very early in life. His father bought him a title of nobility before he was 30 years old, and soon after he purchased a country estate. This did not, however, distract him from his interest in science. In 1768, at the age of 25, he presented a paper on the analysis of water samples, and as a result he was admitted to the Academy as associate chemist. He became director of the Academy in 1785 and treasurer in 1791.
Lavoisier is rightly considered the founder of modern chemistry. He began by disproving the Aristotelian concept of earth, air, fire and water through careful quantitative experimentation. Chemists generally believed at the time that water converts to earth through repeated distillation. Through precise weighing Lavoisier proved that the residue found after several distillations stems from the glass vessel of the distilling apparatus.
His next step was a series of experiments on combustion. Lavoisier proved that sulfur and phosphorus increased in weight during burning, while litharge (lead oxide PbO) heated with charcoal turned into metallic lead that weighed less than the original quantity of litharge. In his book Opuscules physiques et chimiques of 1774 he explained the gain or loss of weight through addition or loss of "air."
In the same year the chemist Joseph Priestley reported on experiments in which he had isolated that part of the air that is responsible for combustion, Lavoisier realized that air is a combination of different gases. He identified "respirable air" (oxygen) and "nonvital air" (nitrogen) and showed that birds lived longer in respirable air. He also showed that respirable air combined with carbon to produce "fixed air" (carbon dioxide). Lavoisier assigned the name "oxygen" (acid producer) to the respirable component of air.
Lavoisier's discovery that air was composed of different gases and that these gases can react with others changed the entire outlook of chemistry. Scientists concentrated on understanding the composition of man substances. in 1783 Lavoisier discovered what the English chemist Henry Cavendish had discovered shortly before him, that water is composed of two gases. By decomposing it into hydrogen and oxygen he collected "inflammable air" (hydrogen) for the purpose of filling balloons for lighter-than-air flight.
In 1789 Lavoisier published his Traité élémentaire de chimie (Elementary Treatise of Chemistry) and founded the chemical journal Annales de chimie. He collaborated with the mathematician Pierre-Simon Laplace on thermochemistry and the different states (solid, liquid and gas) of matter. He studied heat in biological processes such as fermentation and animal heat and concluded that respiration was a type of oxydation reaction similar to the burning of carbon (both produce carbon dioxide).
Like most other scientists of the time Lavoisier took an active part in the administration of the government. in 1768 he joined the Ferme Générale, the state tax collecting agency, where he showed outstanding administrative ability. In 1775 he was appointed director of the gunpowder administration and moved into the Arsenal of Paris. He managed to keep one day of the week free from administrative duties to perform experiments in the laboratory that he had built in the Arsenal, and his apartment became a regular meeting place for scientists.
In 1787, two years before the Revolution, the landowner Lavoisier was elected a member of the provincial assembly of Orléans. When the revolution broke out he was elected to the commune of Paris. As a political liberal he was not averse to reform and supported the revolution through participation in various committees, such as the planning group for the national finances and agriculture and the committee for the introduction of the metric system.
But his high profile in the administration of the old regime placed Lavoisier under increasing suspicion. The Ferme Générale was abolished in 1971, and three years later the revolutionary government ordered the arrest of its former members. Lavoisier and 27 others were sent to the guillotine and buried in a common grave.
Reference
Duveen, D. I. (1995) Antoine-Laurent Lavoisier. Encyclopaedia Britannica 15th ed.

Title page of Lavoisier's Traité élémentaire de chimie .
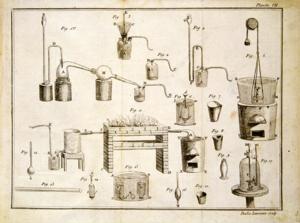
A double page of illustrations from Lavoisier's Traité élémentaire de chimie . Staatsbibliothek zu Berlin, Preussischer Kulturbesitz, Rara-Sammlung.
home
 Lavoisier was the son of a parliamentary counsel, who could afford to give his son an excellent classical education in language, literature, and philosophy combined with best available training in the sciences, including mathematics, astronomy, chemistry, and botany. Lavoisier entered university to study law and received his licence to practice in 1764. His main interest, however, was science. In 1766 he submitted an entry to the competition organized by the Academy of Sciences on the best ways to illuminate a large town and received a gold medal.
Lavoisier was the son of a parliamentary counsel, who could afford to give his son an excellent classical education in language, literature, and philosophy combined with best available training in the sciences, including mathematics, astronomy, chemistry, and botany. Lavoisier entered university to study law and received his licence to practice in 1764. His main interest, however, was science. In 1766 he submitted an entry to the competition organized by the Academy of Sciences on the best ways to illuminate a large town and received a gold medal.
