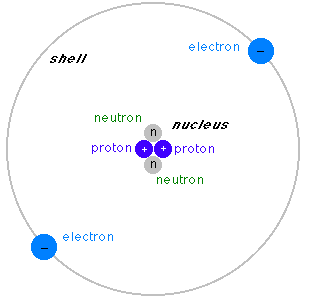

A model of an atom. In this example the nucleus consists of two protons and two neutrons. Real atoms can have as few as a single proton (the Hydrogen atom) or more than 100. Some atoms do not have any neutrons, others have a large number.
This example of an atom has a single shell that carries two electrons. Real atoms can have several shells. In each shell electrons occupy orbits, called orbitals in quantum physics. Not more than two electrons can occupy the same orbitals, and the number of orbitals on a shell cannot be larger than the number of the shell (the shells are counted from the nucleus outward).
The mass of protons and neutrons is vastly larger than the mass of electrons. As a result the atomic weight (the measure of an atom's mass) is very close to the total number of its protons and neutrons.